Developing new pre-clinical models of genetic DEE (developmental and epileptic encephalopathy)
We are delighted to have been awarded a Project Grant from the Neurological Foundation which will enable us to develop our tadpole model to study de novo variants (ie not inherited from parents) that cause these devastating and largely untreatable early onset seizure disorders. This project is being run in collaboration with fellow Zoology researcher Dr Paul Szyszka and Prof Lynette Sadleir from Paediatrics and Child Health, University of Otago Wellington.
DEE are a diverse group of genetic disorders characterised by the onset of difficult-to-treat seizures in the first year of life, leading to developmental regression or stalling. The clinical "gene hunters" - like Prof Sadleir- have identified over 100 genes linked to DEE, but functional studies to determine why variants in these genes lead to seizures are failing to keep pace. Xenopus tadpoles make good models for human disorders, and we will be developing methods to model NZ patient specific variants in 6 genes using CRISPR/Cas9 or mRNA overexpression, analysing seizures by video capture and electrophysiology, and developing models for rapid drug screening.
ARF Dr. Sandesh Panthi is taking the lead on this project. Sandesh joins us from the nearby Department of Anatomy, where he undertook a PhD project with A/Prof Beulah Leitch.
The microbiome and regeneration
 In 2020, we received funding from the Royal Society of New Zealand Marsden Fund to develop our preliminary research into how the skin microbiome of Xenopus tadpoles can be manipulated and how it affects regenerative outcomes, in collaboration with Dr Xochitl Morgan (Microbiology and Immunology) and Dr Robert Day (Biochemistry). Dr Phoebe Chapman is our postdoctoral fellow on this project, and Dan Hudson has been working as a research assistant. The project also involved genetics student Campbell Gilbert, who recently completed his MSc degree. Campbell was able to show that LPS extracted from commensal and non commensal bacteria had similar effects in increasing the chances of tail regeneration. A preprint of our latest submission featuring Campbell's work and the work of Thomas Devine (microbiology BSc Hons) was published in the journal Wound Repair and Regeneration and can be accessed here: Chapman et al, 2022.
In 2020, we received funding from the Royal Society of New Zealand Marsden Fund to develop our preliminary research into how the skin microbiome of Xenopus tadpoles can be manipulated and how it affects regenerative outcomes, in collaboration with Dr Xochitl Morgan (Microbiology and Immunology) and Dr Robert Day (Biochemistry). Dr Phoebe Chapman is our postdoctoral fellow on this project, and Dan Hudson has been working as a research assistant. The project also involved genetics student Campbell Gilbert, who recently completed his MSc degree. Campbell was able to show that LPS extracted from commensal and non commensal bacteria had similar effects in increasing the chances of tail regeneration. A preprint of our latest submission featuring Campbell's work and the work of Thomas Devine (microbiology BSc Hons) was published in the journal Wound Repair and Regeneration and can be accessed here: Chapman et al, 2022.
Why amphibians can regenerate their limbs and tails (sometimes)
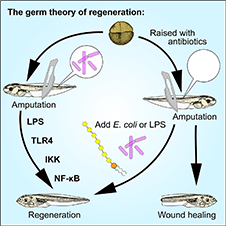 Amphibians often have regenerative capabilities we can only dream of. However, unlike salamanders, frogs can only regenerate effectively some of the time. For tails, which contain spinal cord- of obvious significance for regeneration study- there is a short period of the tadpoles life when the regeneration fails, for unknown reasons. For limbs, the older the tadpole or froglet, the less effective regeneration becomes (Beck et al 2003; Beck et al 2006; Pearl et al 2008; Beck 2012). Thomas Bishop, who completed his PhD in 2018, has uncovered a pathway linking stress factors to the cell cycle, careful control of which could be the key to unlocking regeneration in humans. This work has now been published in iScience as Bishop and Beck, 2021 "Bacterial lipopolysaccharides can initiate regeneration of the Xenopus tadpole tail."
Amphibians often have regenerative capabilities we can only dream of. However, unlike salamanders, frogs can only regenerate effectively some of the time. For tails, which contain spinal cord- of obvious significance for regeneration study- there is a short period of the tadpoles life when the regeneration fails, for unknown reasons. For limbs, the older the tadpole or froglet, the less effective regeneration becomes (Beck et al 2003; Beck et al 2006; Pearl et al 2008; Beck 2012). Thomas Bishop, who completed his PhD in 2018, has uncovered a pathway linking stress factors to the cell cycle, careful control of which could be the key to unlocking regeneration in humans. This work has now been published in iScience as Bishop and Beck, 2021 "Bacterial lipopolysaccharides can initiate regeneration of the Xenopus tadpole tail."
Spinal cord regeneration
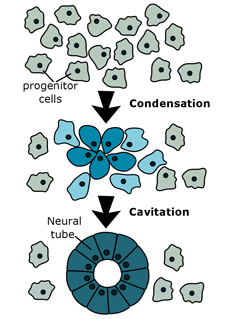
Could tail bud development hold the key to unlocking spinal cord regeneration? The vertebrate central nervous system (CNS) is initially constructed by means of two distinct mechanisms. Secondary neurulation, or spontaneous neural tube assembly (SNTA) forms the CNS arising from the developing vertebrate tail bud, a process that re-occurs in amphibian spinal cord regeneration.
We can also induce this process in animal cap explants, which are comprised of primative, suggestible cells, by activating two cell signalling pathways: wnt and notch, simultaneously (Beck and Slack, 1999). We are undertaking a comparative analysis of transcriptomes of tissues undergoing SNTA to determine whether the processes are the same in development, regeneration, and cultured explants. We are also in the process of collecting timeline data from stage 8 to 37 in explants undergoing SNTA.
Limb development
We have been developing Xenopus as a model for limb development, more commonly studied in other vertebrates such as mice and chickens (Keenan and Beck 2016). We have been investigating the role of gradients of retinoic acid (derived from Vitamin A) in this process (McEwan et al 2011), and are using transcriptome data to identify possible downstream targets.
as mice and chickens (Keenan and Beck 2016). We have been investigating the role of gradients of retinoic acid (derived from Vitamin A) in this process (McEwan et al 2011), and are using transcriptome data to identify possible downstream targets.
Recently we showed that overexpressing the BMP inhibitor Grem1 in transgenic frogs during early limb bud development creates branched and even multiple limbs. This suggests a role for BMP signalling (of which Grem1 is one of several secreted inhibitors) in preventing limb buds from splitting during development (Wang et al 2016). Our latest work on the early limb bud proximodistal axis transcriptome has now been published in Developmental Dynamics (Hudson et al 2022)
 Modelling human disease in Xenopus
Modelling human disease in Xenopus
In a recent exciting collaboration with Associate Professor Louise Bicknell from the department of Pathology, we are developing animal models for the rare congenital disease Meier-Gorlin syndrome, which affects skeletal development and growth. Associate Professor Bicknell has identified mutations in candidate genes from human patients, and we will be developing gene editing in the frog model in order to learn how these mutations give rise to the phenotype.
Xenopus skeletal development and structure is remarkable similar to that of humans, and the external development of the embryos makes them a good target for gene editing.
The Grem1 gene and female fertility
 The grem1 gene codes for a protein that inhibits the action of multiple members of a growth factor family known as bone morphogenetic proteins (BMPs). Certain members of this BMP family can regulate both fecundity and fertility in mammals via their action on oocyte (egg) development. Female transgenic frogs carrying grem1 under the control of a stress inducible Hsp70 promoter (“G” frogs) do not produce many offspring, whereas male carriers have normal numbers of offspring.
The grem1 gene codes for a protein that inhibits the action of multiple members of a growth factor family known as bone morphogenetic proteins (BMPs). Certain members of this BMP family can regulate both fecundity and fertility in mammals via their action on oocyte (egg) development. Female transgenic frogs carrying grem1 under the control of a stress inducible Hsp70 promoter (“G” frogs) do not produce many offspring, whereas male carriers have normal numbers of offspring.
We have used a variety of approaches to investigate the development of the oocytes in a number of grem1 transgenic females. It turns out that the failure of early embryogenesis is due to down regulation if endogenous grem1 in oocytes and ovaries from G females, which looks like it arises from transgene supression. This research, which is now published, could enable future development of a new target for regulating fertility (Beck et al 2018).
Lens development and regeneration
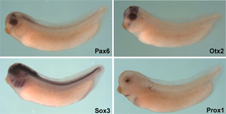 In Xenopus tadpoles, it is possible to study both development and regeneration of parts of the eye. Both the lens and the retina can regenerate. In the case of the lens, removal results in an intriguing process whereby the overlying cornea cells transdifferentiate- that is, stop being corneal cells and turn into lens cells- eventually resulting in the formation of a whole new lens.
In Xenopus tadpoles, it is possible to study both development and regeneration of parts of the eye. Both the lens and the retina can regenerate. In the case of the lens, removal results in an intriguing process whereby the overlying cornea cells transdifferentiate- that is, stop being corneal cells and turn into lens cells- eventually resulting in the formation of a whole new lens.
Our transcriptome analysis of cornea to lens transdifferention implicated wnt signalling in the process, but recently it has been shown that it is inhibition of wnt signalling, rather than activation, that is required (Day and Beck, 2011).